Listen to the article
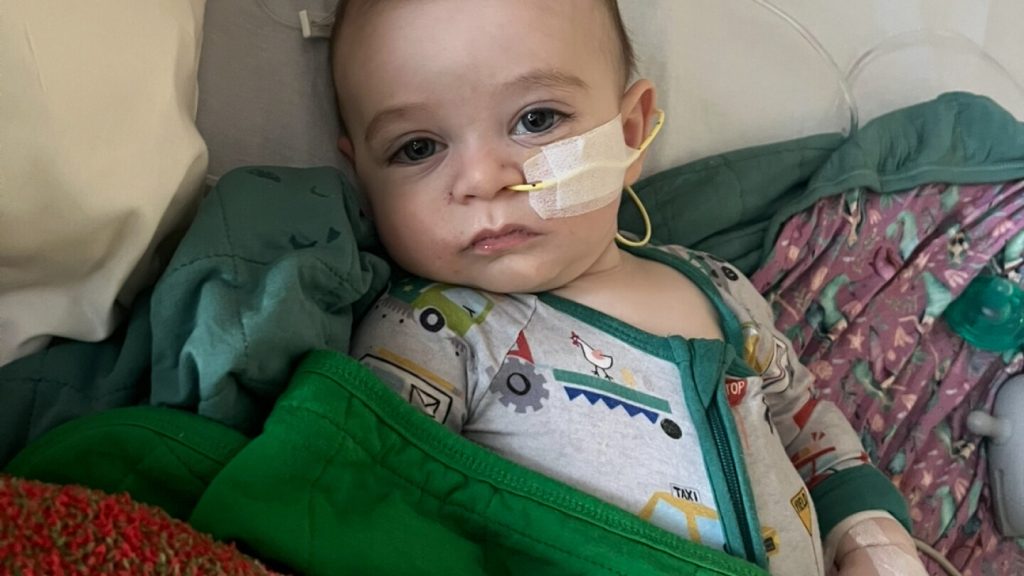
Health officials are currently investigating more than 30 cases of infant botulism linked to ByHeart baby formula since August, but some parents claim their children were sickened by the same product months before the current outbreak was identified.
California public health officials confirmed Friday that six babies in the state who consumed ByHeart formula were treated for botulism between November 2024 and June 2025—up to nine months before the current multi-state outbreak. However, officials stated there was “not enough evidence to immediately suspect a common source” during those earlier cases and that they cannot definitively connect pre-August cases to the current outbreak.
Several families have come forward with accounts of infants who fell ill after consuming the formula earlier in the year. Amy Mazziotti of Burbank, California, reported her then-5-month-old son Hank was diagnosed with botulism in March, weeks after he began drinking ByHeart formula. Similarly, Katie Connolly from Lafayette, California, said her 8-month-old daughter was hospitalized in April with botulism after being fed the same brand.
“What I want to know is why did the cases beginning in August flag an investigation, but the cases that began in March did not?” Connolly asked.
The company recalled all of its products nationwide on November 11 as the outbreak expanded. ByHeart officials have since confirmed that laboratory tests of previously unopened formula found some samples contaminated with the bacterium that causes infant botulism.
Bill Marler, a Seattle-based food safety attorney representing affected families, told The Associated Press that at least three additional pre-outbreak cases involved babies who consumed ByHeart formula and developed botulism. One case dates back to December 2024, with others occurring in spring 2025.
Dr. Jennifer Cope, a CDC scientist leading the investigation, acknowledged awareness of the earlier cases but explained that current efforts are focused on understanding the unusual surge of infections documented since August 1.
“That doesn’t mean that they’re not necessarily part of this,” Cope said. “It’s just that right now, we’re focusing on this large increase.”
She noted that the passage of time and lack of preserved product samples from earlier cases makes it difficult to definitively link them to the current outbreak. Parents who used the formula months ago likely didn’t record lot numbers or keep empty containers, complicating investigation efforts.
Health officials emphasized that the connection between ByHeart and infant botulism only became apparent in recent weeks. Before this outbreak, no powdered infant formula in the U.S. had tested positive for the bacterium that causes botulism, according to California health officials. Early case numbers also fell within expected ranges, and tests on an open container of formula fed to a sick baby in spring failed to detect the pathogen.
The investigation gained momentum when cases began appearing on the East Coast from August through October involving a toxin type rarely detected in that region. Officials noted a pattern of cases in very young infants and a disproportionate number involving ByHeart formula, which represents less than 1 percent of infant formula sales in the United States.
The breakthrough came earlier this month when a sample from a container of ByHeart formula fed to a sick infant tested positive for the bacterium, prompting notification to the CDC, FDA, and the public.
Infant botulism is rare, with fewer than 200 cases reported annually in the U.S. The illness occurs when babies ingest bacterial spores that germinate in the gut and produce a toxin. Since the bacterium exists naturally in soil and water, the source often remains unknown in isolated cases.
Food safety experts, including former FDA deputy commissioner Frank Yiannas, argue that earlier cases should be included in the outbreak investigation if the babies consumed ByHeart formula and were treated for botulism. “Absolutely, yes, they should be included,” Yiannas said. “Why wouldn’t they be included?”
Sandra Eskin, chief executive of STOP Foodborne Illness, agreed, noting, “This outbreak is traumatic for parents. They may have fed their newborns and infants a product they assumed was safe. And now they’re dealing with hospitalization and serious illness of their babies.”
While Connolly’s and Mazziotti’s babies are improving, they report lingering effects from their bouts with botulism, which can cause constipation, poor feeding, muscle weakness, and other symptoms.
“We deserve to know the data that can help us understand how our babies got sick,” Connolly said.
Fact Checker
Verify the accuracy of this article using The Disinformation Commission analysis and real-time sources.



9 Comments
This is a very concerning situation. If the earlier cases were not properly investigated, it raises serious questions about the company’s quality control and the regulators’ oversight. Infant health and safety should be the top priority.
This is very concerning. If there were earlier cases of infant illness linked to this formula, the company and regulators should have investigated more thoroughly. Proper safety protocols need to be in place to protect vulnerable babies.
I agree. It’s unacceptable that the earlier cases were not properly investigated and connected to the current outbreak. The health of infants should be the top priority.
Botulism is extremely serious, especially for young infants. I hope the authorities can get to the bottom of this and determine the root cause, whether it’s contamination, manufacturing issues, or something else. The safety of baby formula is critical.
Absolutely. The families affected must be devastated. Rigorous testing and quality control measures need to be in place to prevent these kinds of outbreaks from happening.
This raises a lot of questions. Why weren’t the earlier cases properly investigated? What went wrong in the manufacturing or distribution process? There needs to be full transparency to ensure this doesn’t happen again.
Agreed. The public deserves answers, and the responsible parties must be held accountable. The health and safety of infants should be the top priority for everyone involved.
It’s alarming that there were potentially months of earlier cases that went undetected. The company and regulators need to thoroughly investigate the entire timeline and supply chain to determine what went wrong. Proper safeguards must be put in place.
Absolutely. This is a serious breach of public trust. Babies’ lives were put at risk, and that is unacceptable. A full, transparent investigation is crucial to prevent this from happening again.