Listen to the article
A promising cancer treatment approach using bee venom requires further research before clinical application, scientists caution.
Recent laboratory research on honeybee venom has sparked widespread social media claims that it can “destroy 100% of breast cancer in less than 60 minutes.” However, oncology experts warn that these statements significantly overstate the current scientific evidence and could potentially mislead patients seeking treatment options.
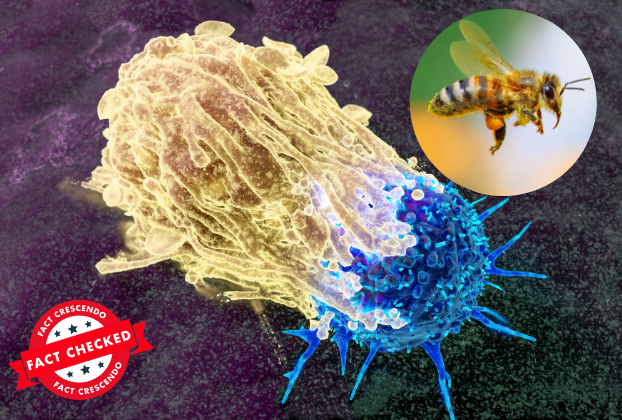
The viral claims stem from a 2020 peer-reviewed study published in npj Precision Oncology, which examined how honeybee venom and its key component, melittin, affect breast cancer cells in controlled laboratory conditions. The research did demonstrate promising results – at specific concentrations, melittin disrupted cancer cell membranes and induced cell death in aggressive breast cancer subtypes, particularly triple-negative and HER2-enriched cancer cells.
Under carefully controlled laboratory settings, researchers observed complete cell death within approximately 60 minutes for certain cancer cell lines. The mechanism involves melittin forming pores in cancer cell membranes and interfering with critical growth pathways like EGFR and HER2. Importantly, the study noted some selectivity, with normal cells being less affected at certain doses.
However, medical experts emphasize that these laboratory findings represent only the earliest stage of potential drug development. Dr. Ciara O’Sullivan, an oncologist not involved in the original research, explains: “The gap between killing cancer cells in a petri dish and developing an effective cancer treatment for humans is enormous. What works in isolated cells often fails when introduced to the complex human body.”
Several crucial limitations exist in translating these findings to clinical treatment. The study focused exclusively on specific breast cancer cell lines, primarily aggressive subtypes, making it impossible to generalize results across all forms of breast cancer. Additionally, the concentration required to achieve rapid cell death in the laboratory setting may not translate to a safe or effective dose in humans.
Bee venom presents significant safety challenges as a potential therapeutic agent. It contains numerous biologically active compounds that can be toxic, and melittin itself disrupts cell membranes broadly – not just in cancer cells – raising concerns about damage to healthy tissues. Bee venom is also a known allergen that can trigger severe immune reactions, including potentially fatal anaphylaxis in susceptible individuals.
The research represents what scientists call “preclinical” work – the preliminary exploration phase that comes before animal studies and the multi-phase clinical trials required to establish safety and efficacy in humans. None of these essential steps have been completed for bee venom as a cancer treatment.
“These findings are scientifically interesting and highlight melittin as a compound worthy of further investigation,” notes Dr. Jennifer Walsh, a cancer researcher at Pacific Medical University. “But characterizing this as a proven cancer treatment is premature and potentially dangerous if it leads patients to delay established treatments.”
Major regulatory authorities including the U.S. Food and Drug Administration and the European Medicines Agency have not approved honeybee venom or melittin for cancer treatment, as there is no clinical evidence from human trials demonstrating safety or effectiveness.
Healthcare organizations emphasize that established cancer treatments – surgery, chemotherapy, radiation therapy, and newer targeted and immunotherapy approaches – have undergone rigorous clinical testing to ensure both safety and efficacy. In contrast, bee venom research remains at the experimental stage.
While the early laboratory findings on melittin show promise for potential future drug development, experts caution that misrepresenting preliminary research as a proven treatment can pose real risks to patients who might delay seeking evidence-based care.
As research continues, scientists will explore whether melittin-based therapies could someday be developed into safe and effective cancer treatments, but that day remains well into the future pending years of additional research and clinical trials.
Fact Checker
Verify the accuracy of this article using The Disinformation Commission analysis and real-time sources.



20 Comments
The cost guidance is better than expected. If they deliver, the stock could rerate.
Exploration results look promising, but permitting will be the key risk.
Interesting update on Fact Check: False Claims About Bee Venom’s Effect on Breast Cancer. Curious how the grades will trend next quarter.
Good point. Watching costs and grades closely.
Good point. Watching costs and grades closely.
Nice to see insider buying—usually a good signal in this space.
Good point. Watching costs and grades closely.
I like the balance sheet here—less leverage than peers.
Good point. Watching costs and grades closely.
Interesting update on Fact Check: False Claims About Bee Venom’s Effect on Breast Cancer. Curious how the grades will trend next quarter.
Good point. Watching costs and grades closely.
Good point. Watching costs and grades closely.
Exploration results look promising, but permitting will be the key risk.
Good point. Watching costs and grades closely.
Uranium names keep pushing higher—supply still tight into 2026.
Good point. Watching costs and grades closely.
If AISC keeps dropping, this becomes investable for me.
Good point. Watching costs and grades closely.
Good point. Watching costs and grades closely.
I like the balance sheet here—less leverage than peers.